Abstract
KMT2A partial tandem duplication (KMT2A-PTD), characterized by a large internal duplication spanning 6-8 exons, has been documented in adult AML with a prevalence of 3-10% and is associated with poor outcomes. Despite the high prevalence of KTM2A fusions and known associated outcomes, prevalence and clinical implications of KMT2A-PTD has not been well characterized in childhood AML. We interrogated the transcriptome and associated clinical and genomic data from pediatric AML patients treated on COG AAML1031 to define the prevalence of KMT2A-PTD, frequency of co-occurring genetic mutations and outcomes associated with this variant.
Ribo-depleted RNA seq data from 1,294 patients with AML (age 0-29 years) enrolled on COG AAML1031 with available cytogenetic, molecular, and clinical data were utilized. Gene fusions, internal tandem duplications (ITD) and partial tandem duplications (PTD) were detected by Cicero. Conventional karyotyping and mutational analysis including NGS were used to determine additional cytogenetic and molecular abnormalities. Response was measured by 5-year overall survival (OS), event-free survival (EFS), disease-free survival (DFS) and relapse risk (RR).
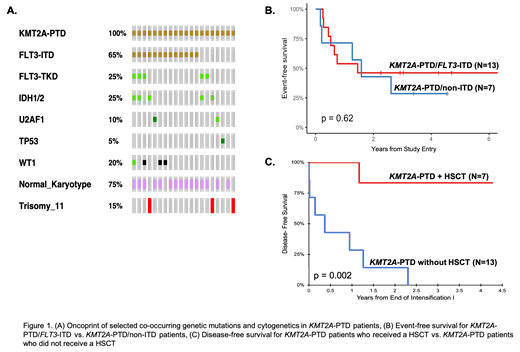
Of the 1,294 patients studied, KMT2A-PTD was identified in 20 patients (1.5%). Evaluation of karyotype demonstrated striking paucity of karyotypic alterations with 75% with normal karyotype; 3 cases (15%) of trisomy 11 were identified. In contrast to KMT2A-rearranged (KMT2A-r) AML, which has the highest prevalence in young patients (median age 3.3 years), the median age with KMT2A-PTD was 13.4 years (p<0.0001). Further analysis demonstrated a high rate of co-occurrence of FLT3 mutations; 15 patients (75%) had FLT3-ITD (N=13; 65%) and/or FLT3 activating mutations (N=5; 25%). The rate of co-occurring FLT3-ITD was significantly higher in the KMT2A-PTD cohort compared to patients without KMT2A-PTD (65% vs. 19%, p<0.001). Prevalence of KMT2A-PTD in patients with FLT3-ITD was 5.7%. Additional co-occurring genetic mutations were identified in WT1 (20%) and the RAS pathway. There was a striking enrichment of mutations that are commonly associated with AML in older adults with 8/20 patients (40%) harboring mutations in IDH1/IDH2, U2AF1, and TP53, far in excess of what is seen in other pediatric AML patients (Figure 1A).
We evaluated the significance of KMT2A-PTD on clinical outcome. Patients with KMT2A-PTD had a poor response to induction therapy with a morphologic CR rate of 45% and rate of residual disease (MRD) positivity by flow cytometry after initial course of therapy was 40%. Additionally, high rates of relapse were noted for patients with KMT2A-PTD with a RR of 53%. EFS for patients with and without KMT2A-PTD was 39% vs. 46%, respectively (p=0.54) with a corresponding OS of 58% and 64% (p=0.61). Outcome analysis for the KMT2A-PTD cohort demonstrated dual KMT2A-PTD/FLT3-ITD patients had an EFS of 46% compared to 29% for KMT2A-PTD/non-ITD patients (p=0.62, Figure 1B). We inquired whether hematopoietic stem cell transplant (HSCT) may modify outcomes in patients with KMT2A-PTD. Of the 20 patients with KMT2A-PTD, 7 (35%) proceeded to HSCT in CR1, with the indication in 6 of 7 patients due to FLT3-ITD high allelic ratio (HAR, ≥0.4). Stem cell transplant recipients had a DFS of 83% vs. 0% for those who continued on protocol chemotherapy (p=0.002, Figure 1C) with corresponding OS of 100% vs. 43%, respectively (p=0.05).
In this large cohort of childhood AML patients treated on AAML1031, we identified a small subset of patients with KMT2A-PTD and show the high prevalence of co-occurring FLT3 mutations. Although KMT2A-PTD patients with and without FLT3-ITD had similar outcomes, the cohort who received HSCT in CR1 (most with FLT3-ITD) experienced improved outcomes compared to the rest of the cohort, suggesting that intensification of therapy may benefit this group of patients overall. Further research into KMT2A-PTD in pediatric AML will guide future risk-adapted therapy and enhance understanding of biologic implications of this lesion, including whether altered KMT2A may serve as a therapeutic target as it may for KMT2A-r AML.
Pollard: Syndax: Membership on an entity's Board of Directors or advisory committees; Kura Oncology: Membership on an entity's Board of Directors or advisory committees.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal